November 27, 2023
The term “precision medicine” refers to a broad range of medical treatments that are based on the individual characteristics of the patient. Precision medicine recognizes that each person’s characteristics, including their genetic profile, may determine their likelihood of disease development and/or a successful response to treatment. Scientists agree that precision medicine has great potential for providing high value care, improving outcomes and reducing treatment costs for some patients.
Genetic science is the key driver of progress in precision medicine. Genetic testing and gene therapy target different outcomes: genetic testing is aimed at identifying the potential for disease or diagnosing a genetic mutation that causes it, as well as informing treatment protocol; gene therapies focus on treating disease that has already been diagnosed.
But, for all their potential, genetic testing and therapies are complicated, in some cases expensive and difficult to track with respect to utilization and appropriateness, presenting employers with unique challenges. This section of the guide summarizes employer considerations and recommendations that may help with the evaluation of the impact of genetic testing and gene therapies on benefit costs, programmatic design and evidence-based coverage decisions, as well as improve the patient experience.
In Simple Terms:
Precision Medicine: Medical care that focuses on identifying approaches that would effectively treat a particular patient based on their individual genetic, environmental and lifestyle factors.
Genome/Genomics: The complete set of genes in an organism and the study of these genes.
Genetics: The study of heredity and variation in characteristics of people based on their heredity.
Genes: The basic unit of heredity transferred from parents to offspring, present in all cells.
Genetic Testing: Analysis of a particular gene, or set of genes, that may indicate the presence of or increased likelihood of developing a disease.
Gene Therapy: Medical technique involving transplanting genetic material into human cells in order to replace or correct for missing or defective genes.
Pharmacogenomic Tests: Genetic testing to determine how an individual will respond to a given medication or therapy based on their genetic makeup.
Although precision medicine can improve outcomes and reduce costs for individual patients, the overall impact of genetic testing and gene therapies on health care costs will likely continue to grow. This assessment is based on the explosion in the number of tests becoming available and increased focus on gene therapies by drug companies. Employers find the prospect of paying millions of dollars for these gene therapies challenging and unsustainable for the overall viability of their health plans. The 2024 Large Employer Health Care Strategy Survey indicates that many employers are either very concerned (53%) or concerned (26%) about the patient and plan affordability of gene and cell-based therapies in the pipeline.5
While genomics is an exciting and growing branch of medicine, there are new considerations that employers should be aware of. Improved employer understanding of genetic testing and the treatment landscape can result in better coverage decisions.
Based on our research, we have summarized the key employer considerations and recommendations:
Genetic Testing Has Clinical Utility, But It Should Not Be Used to Satisfy Patient Curiosity
Genetic testing, when used correctly, can help diagnose a disease, determine the appropriateness of treatment or indicate the likelihood of developing the disease. The test results may also impact the patient’s course of preventive screenings or, in some cases, cause patients to take preventive action. But genetic testing doesn’t always provide definite answers: instead, the results of some tests can create significant anxiety and fear of developing a specific illness, often with few actionable suggestions or clinical treatments available to prevent it.
Many of the genetic tests available do not have high-quality, unbiased research showing their effectiveness. In addition, many health care providers weren’t trained to use or interpret genetic testing, so even the most accurate test wouldn’t help them make better diagnosis or treatment decisions. False positives can lead to additional and expensive testing and treatment. Therefore, consider genetic tests’ clinical utility when making coverage decisions.
Use Prior Authorizations…Efficiently
Prior authorizations are an important tool in ensuring appropriate utilization of genetic testing but should be performed by professionals experienced in the area of genetics and automated whenever possible to avoid delays. Patients, providers and even expert geneticists, who use genetic testing appropriately, often run into roadblocks with health plan prior authorization processes, in part because claims are submitted via the lab testing company rather than the physician.
Precision Medicine Coverage Decisions Are Complex
The Business Group’s 2024 Large Employer Health Care Strategy Survey looked at various aspects of employer’s coverage of precision medicine. Fifty-five percent of large employers will have at least one aspect of precision medicine coverage in place in 2024 and that number will likely climb in the future.
Some actions employers who want to play a more active role in genetic testing coverage decisions may include:
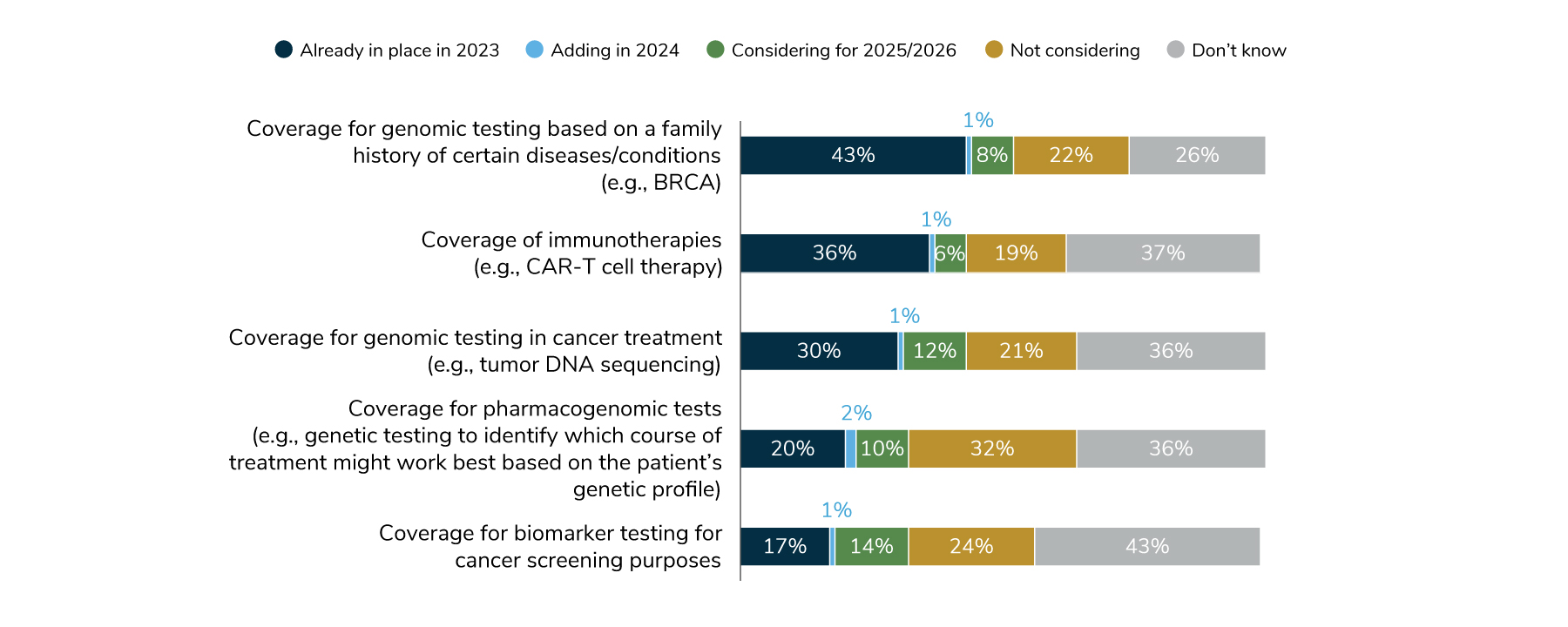
Source: Business Group on Health. 2024 Large Employer Health Care Strategy Survey. August 22, 2023. https://www.businessgrouphealth.org/resources/2024-large-employer-health-care-strategy-survey-health-and-pharmacy-plan-design. Accessed September 21, 2023.
These complex coverage decisions are often made based on individual tests or therapies and are subject to utilization management decisions that take into account their applicability for a particular patient.5 Employer sponsored Health plans typically pay for genetic counseling and, in many cases, genetic testing when it is recommended by a doctor and deemed medically necessary.
Some actions employers who want to play a more active role in genetic testing coverage decisions may consider include:
- Discussing with their health plans how they make coverage decisions for emerging genetic tests and ensuring that such decisions are based on the latest evidence and made in consultation with genetic experts across multiple subspecialties;
- Understanding the frequency with which such decisions are being made. The rapid pace of clinical development in genetic testing requires much higher levels of vigilance and coverage changes to ensure that coverage policies are flexible enough to serve those in clinical need.
- Utilization management programs may have to be deployed to assist in the determination of clinical need and assessment of utilization appropriateness, as well as to prevent waste and harm of unnecessary testing;
- Asking their health plans to require genetic counseling prior to prescribing tests, a practice already adopted by major health plans. In 2013, Cigna became the first insurer in the U.S. to require its members to receive genetic counseling before it would pay for them to undergo genetic tests for breast, ovarian or colorectal cancers; and
- Requesting that their partners report on projected increases in spending resulting from any coverage decision changes made and the estimated number of members that may benefit from such increased coverage.
Cost Is a Factor for Genetic Testing—And You Don’t Always Know What You’re Paying For
As of November 2022, a total of 129,624 and 197,779 genetic tests in the US and globally, respectively, including updated versions of previously existing tests, have been made available and submitted to the genetic testing registry..2 With only about 400 CPT codes available to bill for tens of thousands of tests and test panel combinations, it is difficult, if not impossible, to accurately capture the extent or appropriateness of a given genetic test based solely on claims data.6 Furthermore, the costs associated with follow-up testing and potentially unnecessary treatment based on a misapplied genetic test or misinterpreted test results can often exceed the cost of the genetic test itself.
Even employers who have not made an active coverage decision with respect to genetic testing may already be paying for them and can expect the trend to increase. As with any new, rapidly changing and costly area in health care, there is no substitute for timely and thorough analytical review. The availability of tens of thousands of tests but only hundreds of CPT codes means that lab testing companies can obscure the scope of testing they bill for, potentially leaving the benefit open for waste and abuse.
Routine claims reporting can help identify potential areas of opportunity for better utilization management in the area of genetic testing and identify areas of abuse/waste. Such reporting and review should include identification of potential red flags such as:
- Clusters of orders by same provider and inappropriate number of daily visits with the same provider;
- Genetic testing without an appropriate diagnosis code;
- Unnecessarily wide variations in allowed amount for the same test;
- Billed volumes inappropriate for a given procedure code;
- Inappropriate billing code combinations; and
- Cases where follow-up services were not performed after genetic testing.
Employers should also encourage their lab benefit managers (LBMs) or health plans to implement claims edits that prevent overpayment of genetic test claims. Such edits should identify the actual test(s) performed and determine if attributes of the claim violate policy. Edits should consider procedure codes, diagnosis codes, servicing provider, ordering provider and patient demographics.6
While Steering to Quality, Engage with Providers with Expertise in Genetic Testing
Many practicing providers weren’t trained to use newer genetic tests or translate the results for patients. Therefore, employers should ensure that members undergoing genetic testing have access to genetic counselors and should consider deploying utilization management solutions. Possible solutions include ensuring that genetic testing is performed when it is clinically justified and has the potential to improve care or provide diagnostic accuracy.
Even primary care providers who may be in the best position to guide patients to appropriate genetic testing and positive views on the utility of testing, report feeling unprepared to work with patients at high risk for genetic conditions and to interpret the results of testing.7
With genetic testing being most prevalent in oncology and reproductive health, employers engaging with Centers of Excellence or point solutions offering oncology, fertility or maternity services should inquire if their partners provide members and their practicing providers with appropriate access to genetic counseling. Access to experts in genetic science, as well as making use of programs and providers that adopt the latest evidence-based guidelines related to genetic testing and deployment of gene therapies, can influence outcomes and lead to higher efficiency of care.
Gene Therapies May Enhance Patient Care but…They Bring Unprecedented Cost Challenges and Long-term Evidence Is Not Yet Available
Employers should keep in mind that gene therapies are saving and transforming lives right now. Newly approved gene therapies can cure rare diseases, but the initial costs of many of these treatments and associated care top $1 million. Over time, this investment in a potential cure may save the health care system money, however, no long-term evidence is currently available on the effectiveness of these new treatments.
Deploying companion testing prior to prescribing genetic therapy to determine potential efficacy of specialized treatment can help reduce waste of expensive gene therapies, as the cost of testing is much lower in comparison to waste of inappropriate utilization of such therapies.
Other actions employers can take proactively could include:
- Monitoring drug spend under the medical benefit carefully and discussing with carriers any price, discount or rebate negotiations that may be taking place with respect to these therapies;
- Considering proactive conversations with medical carriers regarding coverage of these therapies and understanding available pricing and formulary arrangement options, including whether the carrier may implement an initial exclusion until drug efficacy claims are further proven;
- Monitoring the gene therapy pipeline and encouraging partners to provide notification when certain high-priced therapies are added to coverage;
- Requiring prior authorization and appropriate genetic testing prior to administration of the therapy;
- Ensuring that expensive gene therapies are administered in highly specialized setting; and
- Taking broader action to support reform of drug pricing and addressing supply chain complexities that result in unsustainable growth in prices of individual therapies, putting pressure on the sustainability of coverage of some of the emerging treatments.
A Word About Privacy and Ethical Considerations
Advancement in genetic science has also opened an important and necessary debate about ethics and ethical applications of the tests, as well as data privacy. By law, genetic results cannot be used to discriminate for the purposes of health benefits, but life and disability insurance providers can use them for individual ratings.
Two important considerations employers should be aware of are:
- Direct-to-consumer (DTC) companies typically take measures to protect consumers’ privacy once they submit their DNA samples; measures include storing personal information and genetic data in separate environments in the event of a data breach. Anonymized genetic data are often shared with researchers as well as pharmaceutical companies, which can result in the identification of individuals’ relatives who have never taken a genetic test, if the relative has shared their information with private, non-anonymous DNA databases such as GEDmatch.8 This raises concerns about the degree to which researchers and other databases can “re-identify” significant portions of participants originally thought to be providing anonymous information.
- In the United States, discrimination based on genetic information is prohibited under the Genetic Information Nondiscrimination Act (GINA). However, GINA does not apply to law enforcement use of DTC genetic testing. Furthermore, DTC companies are not bound by the Health Insurance Portability and Accountability Act (HIPAA) privacy laws, illustrating a gap in legal protections that many consumers may not be fully aware of.9 GINA also does not apply to genetic discrimination, defined as differential treatment of individuals by their employers or insurance companies based on their genetic makeup, for types of insurance other than health care (such as life, disability or long-term care insurance).10 Discuss with your life and disability insurance partners their policies related to use and privacy of any genetic testing information.
Member Education and Resources
Employers can play a role in providing patients with access to resources that enable them to make the right decisions about their health, risk levels and appropriateness of genetic testing for their family. As the novelty of genetic testing and the marketing efforts by some testing companies and lab may drive inappropriate utilization, employer communications teams can help manage expectations that genetic testing is not for everyone and that decisions about appropriateness of tests should be made by doctors and counselors.
More Topics
Articles & Guides- 1 | National Institutes of Health. NIH Fact Sheets. Genetic Testing: How It Is Used for healthcare. June 30, 2018. https://archives.nih.gov/asites/report/09-09-2019/report.nih.gov/nihfactsheets/ViewFactSheetef83.html. Accessed December 2, 2020.
- 2 | Halbisen AL, Lu CY. Trends in Availability of Genetic Tests in the United States, 2012-2022. J Pers Med. 2023;13(4):638. Published 2023 Apr 6. doi:10.3390/jpm13040638. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10142561/. Accessed September 20, 2023.
- 3 | U.S. Food & Drug Administration. Approved Cellular and Gene Therapy Products. June 30, 2023. https://www.fda.gov/vaccines-blood-biologics/cellular-gene-therapy-products/approved-cellular-and-gene-therapy-products. Accessed September 20, 2023.
- 4 | Hunt, T. The Cell And Gene Therapy Sector in 2023: A Wave Is Coming - Are We Ready? January 9, 2023. https://invivo.pharmaintelligence.informa.com/IV146781/The-Cell-And-Gene-Therapy-Sector-In-2023-A-Wave-Is-Coming--Are-We-Read. Accessed September 23, 2023.
- 5 | Business Group on Health. 2024 Large Employer Health Care Strategy Survey. August 22, 2023. https://www.businessgrouphealth.org/resources/2024-large-employer-health-care-strategy-survey-intro. Accessed September 20, 2023.
- 6 | Informed DNA. Cost & Delivery Institute Presentation: Genetic Benefits Management: Best Practices. January 22, 2020.
- 7 | Hauser D, Obeng A, Fei K et al. Views of primary care providers on testing patients for genetic risks for common chronic diseases. Health Affairs. 2018 May;37(5):793-800. https://www.healthaffairs.org/doi/10.1377/hlthaff.2017.1548. Accessed December 5, 2020.
- 8 | Brodwin E. After you spit into a tube for a DNA test like 23andMe, experts say you shouldn’t assume your data will stay private forever. Business Insider. February 16, 2019. https://www.businessinsider.com/privacy-security-risks-genetic-testing-23andme-ancestry-dna-2019-2. Accessed December 5, 2020.
- 9 | Hendricks-Sturrup R, Prince AER, Lu CY. Direct-to-consumer genetic testing and potential loopholes in protecting patient privacy and nondiscrimination. JAMA. 2019, 32(19): 1869-1870.https://jamanetwork.com/journals/jama/fullarticle/2731672?widget=personalizedcontent&previousarticle=2706179. Accessed December 5, 2020.
- 10 | Medline Plus. Genetic Testing. September 10, 2020. https://medlineplus.gov/genetics/understanding/testing/. Accessed December 5, 2020.
- 11 | National Society of Genetic Counselors. 2023 Professional Status Survey. 2023. https://www.nsgc.org/Portals/0/2023%20PSS%20Executive%20Summary.pdf. Accessed September 20, 2023.
- 12 | Phillips KA, Deverka PA. The emerging use by commercial payers of third-party lab benefit managers for genetic testing. Health Affairs Blog. October 23, 2019. https://www.healthaffairs.org/do/10.1377/hblog20191021.563154/full/. Accessed December 5, 2020.
- 13 | American Clinical Laboratory Association. Economic Impact of Clinical Labs. https://www.acla.com/economic-impact-of-clinical-labs/. Accessed December 5, 2020.
- 14 | Swetlitz I. Genetic tests ordered by doctors race to market, while ‘direct-to-consumer’ tests hinge on FDA approval. STAT. March 16, 2018. https://www.statnews.com/2018/03/16/genetic-tests-fda-regulation/. Accessed December 5, 2020.
- 15 | Phillips KA, Deverka PA, Hooker GW et al. Genetic testing availability and spending: Where are we now? Where are we going? Health Affairs. 2018 May; 37(5): 710-716. https://www.healthaffairs.org/doi/full/10.1377/hlthaff.2017.1427. Accessed December 5, 2020.
- 16 | Healthcare Fraud Prevention Partnership. White Paper: Genetic Testing: Fraud, Waste & Abuse. July 2020. https://www.cms.gov/files/document/hfpp-genetic-testing-fwa-white-paper.pdf. Accessed December 5, 2020.
- 17 | National Cancer Institute at the National Institutes of Health. Multiple-gene panel test. https://www.cancer.gov/publications/dictionaries/genetics-dictionary/def/multiple-gene-panel-test. Accessed December 5, 2020.
- 18 | American College of Obstetricians and Gynecologists (ACOG). Carrier Screening in the Age of Genomic Science. March 2017. https://www.acog.org/clinical/clinical-guidance/committee-opinion/articles/2017/03/carrier-screening-in-the-age-of-genomic-medicine. Accessed December 5, 2020.
- 19 | United States Preventive Services Task Force (USPSTF). Final Recommendation Statement: BRCA-Related Cancer: Risk Assessment, Genetic Counseling, and Genetic Testing. August 20, 2019. https://www.uspreventiveservicestaskforce.org/Page/Document/RecommendationStatementFinal/brca-related-cancer-risk-assessment-genetic-counseling-and-genetic-testing. Accessed July 8, 2020.
- 20 | Nelson HD, Huffman LH, Fu R, Harris EL, Walker M, Bougatsos C. Genetic risk assessment and BRCA mutation testing for breast and ovarian cancer susceptibility. Agency for Healthcare Research and Quality (US); September 2005. https://pubmed.ncbi.nlm.nih.gov/20722133/. Accessed December 5, 2020.
- 21 | American College of Obstetricians and Gynecologists (ACOG). Carrier Screening FAQs. October 2020. https://www.acog.org/patient-resources/faqs/pregnancy/carrier-screening. Accessed December 5, 2020.
- 22 | Molteni M. 23andMe’s new diabetes test has experts asking who it’s for. Wired. March 10, 2019. https://www.wired.com/story/23andmes-new-diabetes-test-has-experts-asking-who-its-for/. Accessed December 5, 2020.
- 23 | Catalyst for Payment Reform. How-to Guide: Unraveling Genetic Testing Benefits. December 2018. https://www.catalyze.org/product/value-genetic-testing-benefits/. Accessed December 5, 2020.
- 24 | Mayo Clinic. Pharmacogenomics Finding the Right Medication for You. 2013. https://mcforms.mayo.edu/mc1200-mc1299/mc1205-70.pdf. Accessed on December 5, 2020.
- 25 | Aon. Evidence-based Benefit Design Committee Presentation: Introduction to Human Genomics. February 13, 2019.
- 26 | Centers for Disease Control and Prevention (CDC). Mental Health Conditions: Depression and Anxiety. August 6, 2020. https://www.cdc.gov/tobacco/campaign/tips/diseases/depression-anxiety.html. Accessed December 5, 2020.
- 27 | Grohol JM. Gene testing for antidepressants & psychotropics: Not there yet. PsychCentral Blog. December 6, 2019. https://psychcentral.com/blog/gene-testing-for-antidepressants-psychotropics-not-there-yet/. Accessed December 5, 2020.
- 28 | Jørgensen JT, Hersom M. Companion diagnostics—a tool to improve pharmacotherapy. Ann Transl Med. 2016 Dec; 4(24): 482. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5233535/. Accessed December 5, 2020.
- 29 | U.S. Food & Drug Administration (FDA). Companion Diagnostics. December 7, 2018. https://www.fda.gov/medical-devices/vitro-diagnostics/companion-diagnostics. Accessed December 5, 2020.
- 30 | FDA. List of Cleared or Approved Companion Diagnostic Devices (In Vitro and Imaging Tools). November 16, 2020. https://www.fda.gov/medical-devices/vitro-diagnostics/list-cleared-or-approved-companion-diagnostic-devices-vitro-and-imaging-tools. Accessed December 5, 2020.
- 31 | Jørgensen JT. Companion & Complementary Diagnostics: Clinical and Regulatory Perspectives. Dx-Rx Institute Presentation at Workshop on Companion Diagnostics. January 31, 2017. https://biomarkers.dk/fileadmin/user_upload/Editor/Files/Niels/Biomarkers/events/Jan_2017/Jan_170127_-_Companion_and_Complemnetary_Diagnostics.pdf. Accessed December 5, 2020.
- 32 | Levy B. . Clinical Progress and Coverage Policies in Immuno-Oncology. Episode 1: Existing Treatment Landscape of Immunotherapies. July 28, 2016. https://www.ajmc.com/insights/clinical-progress-and-coverage-policies-in-immuno-oncology/distinguishing-between-companion-and-complementary-diagnostic-tests. Accessed December 5, 2020.
- 33 | Vassy JL, Bates D, Murray M. Appropriateness: A key to enabling the use of genomics in clinical practice? The American Journal of Medicine. 2016 Jun; 129(6): 551-553. https://www.amjmed.com/article/S0002-9343(16)30198-X/pdf. Accessed December 5, 2020.
- 34 | CDC. Genomics & Precision Health: Evaluating Genomic Tests. September 20, 2019. https://www.cdc.gov/genomics/gtesting/index.htm. Accessed December 5, 2020.
- 35 | Genomics Education Programme (NHS). What is NIPT? December 18, 2017. https://www.genomicseducation.hee.nhs.uk/blog/what-is-nipt/. Accessed December 5, 2020.
- 36 | National Human Genome Research Institute (NHGRI). Noninvasive Prenatal Genetic Testing. April 17, 2018. https://www.genome.gov/dna-day/15-ways/noninvasive-prenatal-genetic-testing. Accessed December 5, 2020.
- 37 | ACOG. Prenatal Genetic Screening Tests FAQs. October 2020. https://www.acog.org/patient-resources/faqs/pregnancy/prenatal-genetic-screening-tests. Accessed December 5, 2020.
- 38 | Medline Plus. Genetics. August 31, 2020. https://ghr.nlm.nih.gov/primer/newbornscreening/nbscost . Accessed December 8, 2020.
- 39 | Mayo Clinic. Genetic Testing. https://www.mayoclinic.org/tests-procedures/genetic-testing/about/pac-20384827. Accessed December 5, 2020.
- 40 | American Association for the Advancement of Science (AAAS). Research supports expanding insurance coverage of non-invasive prenatal testing. Press Release. October 8, 2019. https://www.eurekalert.org/pub_releases/2019-10/uoca-rse100819.php. Accessed December 5, 2020.
- 41 | United Healthcare. Carrier Testing for Genetic Diseases – Commercial Medical Policy. Effective Date: July 1, 2020. https://www.uhcprovider.com/content/dam/provider/docs/public/policies/comm-medical-drug/carrier-testing-for-genetic-diseases.pdf. Accessed December 5, 2020.
- 42 | Cigna. Genetic Testing for Reproductive Carrier Screening and prenatal Diagnosis – Medical Coverage Policy. Effective Date: December 15, 2019. https://static.cigna.com/assets/chcp/pdf/coveragePolicies/medical/mm_0514_coveragepositioncriteria_genetic_testing_repro_carrier_prenatal.pdf. Accessed December 5, 2020.
- 43 | Rose S, Chun C. The test that saved my sister’s life and my own. Healthline Blog. July 27, 2020. https://www.healthline.com/health/breast-cancer/brca-gene-test-saved-my-life. Accessed December 5, 2020.
- 44 | Filipski KK, Mechanic LE, LongR et al. Pharmacogenomics in oncology care. Front Genet. 2014; 5:73. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3986526/. Accessed December 5, 2020.
- 45 | U.S. National Library of Medicine. ClinicalTrials.gov. 2020. https://clinicaltrials.gov/ct2/results?term=%22gene+therapy%22+OR+%22gene+transfer%22+OR+%22virus+delivery%22&Search=Apply&recrs=b&recrs=a&age_v=&gndr=&type=&rslt=. Accessed December 5, 2020.
- 46 | Blue Cross Blue Shield Association (BCBSA). Evidence-based Benefit Design Committee Presentation: Genetic Testing in Precision Medicine Presentation. February 13, 2019.
- 47 | McCray PB Jr. Difficulties of gene therapy. The Lancet Supplement. 2001 https://www.thelancet.com/pdfs/journals/lancet/PIIS0140673601070325.pdf. Accessed December 5, 2020.
- 48 | Rockoff JD. The million-dollar cancer treatment. Who will pay? The Wall Street Journal. April 26, 2018. https://www.wsj.com/articles/the-million-dollar-cancer-treatment-no-one-knows-how-to-pay-for-1524740401. Accessed December 5, 2020.
- 49 | LifeSciencesIntelligence. The Top 5 Most Expensive FDA-Approved Gene Therapies. May 24, 2023. https://lifesciencesintelligence.com/features/the-top-5-most-expensive-fda-approved-gene-therapies#:~:text=Zolgensma%2C%20and%20Luxturna.-,Hemgenix,as%20congenital%20factor%20IX%20deficiency. Accessed September 20, 2023.
- 50 | FDA. Statement from FDA Commissioner Scott Gottlieb, M.D. and Peter Marks, M.D., Ph.D., Director of the Center for Biologics and Evaluation and Research on new policies to advance development of safe and effective cell and gene therapies. January 15, 2019.https://www.fda.gov/news-events/press-announcements/statement-fda-commissioner-scott-gottlieb-md-and-peter-marks-md-phd-director-center-biologics. Accessed December 5, 2020.
- 51 | American Society of Gene & Cell Therapy. Annual meeting.ASGCT - American Society of Gene & Cell Therapy | ASGCT - American Society of Gene & Cell Therapy. Accessed December 8, 2020.
- 52 | Buckles S. Gene therapy: Potential and pitfalls. Mayo Clinic blog. November 19, 2018. https://individualizedmedicineblog.mayoclinic.org/2018/11/19/gene-therapy-potential-and-pitfalls/. Accessed on December 5, 2020.
- 53 | Business Group on Health. Quick Survey: Genetic Testing Benefit Offering. February 21, 2020. https://www.businessgrouphealth.org/resources/genetic-testing-benefit-offerings-quick-survey. Accessed December 5, 2020.
- 54 | Ugalmugle S, Swain R. Genetic Testing Market Size by Test Type (Predictive Testing, Carrier Testing, Prenatal and New-born Testing, Diagnostic Testing, Pharmacogenomic Testing, Nutrigenomic Testing), By Application (Cancer, Genetic Disease, Cardiovascular Disease), Industry Analysis Report, Regional Outlook, Application Potential, Competitive Market Share & Forecast, 2020 – 2026. Global Market Insights. February 2020. https://www.gminsights.com/industry-analysis/genetic-testing-market. Accessed December 5, 2020.
-
IntroEmployers’ Guide to Precision Medicine: Genetic Testing, Treatments and Implications for Coverage
-
Section 1Genetic Testing and Treatments: Key Challenges and Recommendations for Employers
-
Section 2Genetic Counseling and Benefit Management: New Partnerships Emerge
-
Section 3Genetic Testing Landscape
-
Section 4Key Applications of Genetic Testing in Reproductive Health and Oncology
-
Section 5Gene Therapy: Key Considerations for Employers
-
Section 6Deep Dive into Pharmacogenomics: Deploying Precision Medicine in Drug Prescribing
This content is for members only. Already a member?
Login
![]()
